Abstract
Introduction
Elotuzumab (Elo), an immunostimulatory monoclonal antibody targeting signaling lymphocytic activation molecule F7 (SLAMF7), received marketing approval in Italy for relapsed or refractory multiple myeloma (RRMM), in combination with lenalidomide (R) and dexamethasone (d) (EloRd) in April 2017. Here we report preliminary data of an Italian real-life experience on EloRd as salvage therapy for RRMM patients treated outside of controlled clinical trials.
The primary objectives of this study were to assess the toxicity profile and the efficacy of EloRd administered as salvage therapy in a real-world setting.
Methods
Retrospective data collection received approval from local ethic committee. The cohort included 180 RRMM patients from 28 Italian centers who received at least one cycle of EloRd as salvage treatment between April 2017 and June 2018. Patients were treated with EloRD according to marketing approval as follows: Elo 10 mg/kg i.v. on days 1, 8, 15, and 22 during the first two cycles and then on days 1 and 15 of each following cycle, R 25 mg on days 1 to 21 of each cycle and d at a dose of 40 mg during the week without Elo, and 36 mg on the day of Elo administration. Responsive patients had to reach at least a partial remission (PR).
Results
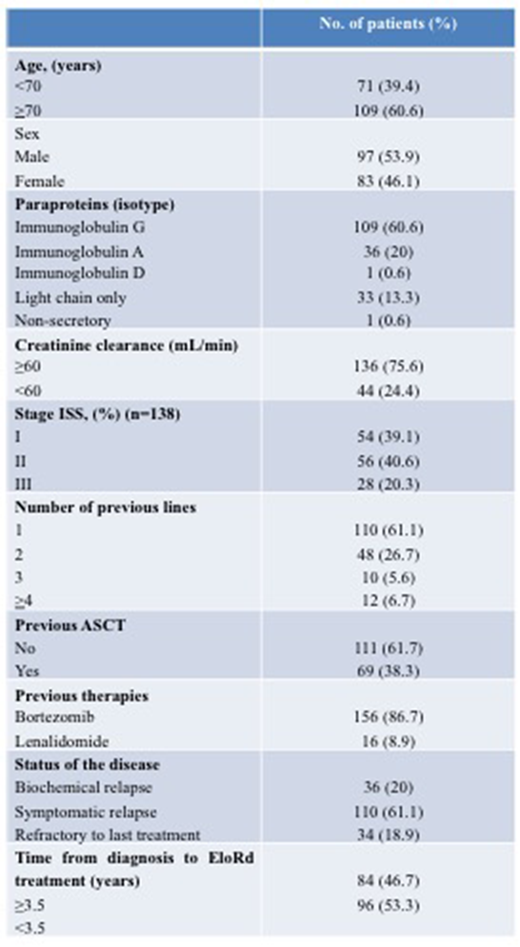
Baseline characteristics are shown in Table 1. Median age of the 180 patients was 72 years (range 48-89 years); 53.9% were males (Table 1). The median number of previous therapy regimens was 1 (range 1-7); 69 patients (38.3%) received a previous autologous stem cell transplantation (ASCT). One hundred and ten patients (61.1%) showed a symptomatic relapse, 36 (20%) a biochemical relapse, and 34 cases (18.9%) were refractory to the last therapy, including bortezomib in 12.8% of patients. Forty-four patients (24.4%) had creatinine clearance ≤60 mL/min. Among the 138 cases evaluable for International Scoring System (ISS), 54 (39.1%) had stage I, 56 (40.6%) stage II, and 28 (20.3%) stage III.
At last data collection (June 2018), 164 cases were evaluable for response. The median number of courses administered so far was 5 (range 1-18). The overall response rate (ORR) was 78%, with 9 complete remissions (CRs) (5.5%) and 49 very good partial remissions (VGPRs) (35.4%). A significant higher ORR was observed in patients who had received only 1 previous line of therapy than those who had received >1 line (64% vs 37.5%; p=0.004). Age (<70 vs ≥70 years), sex, previous ASCT, creatinine clearance (<60 vs ≥60 mL/min), ISS stage (I vs II vs III), time from diagnosis to EloRd start (≥3.5 years vs <3.5 years), status of the disease at EloRd start (biochemical relapse vs symptomatic relapse vs refractory to last therapy) did not significantly impact on the probability of achieving a response. The median time to first response was 1.6 months, while the median time to best response 2.8 months.
After a median follow-up of 6 months (range 1-18 months) 33 patients stopped treatment due to disease progression and 4 due to toxicity (2 cases after 1 cycle for pneumonia, 1 after 1 cycle for dexamethasone-related psychosis, and 1 after 2 cycles for lenalidomide-related severe skin rash). A total of 17 patients died (7 patients from progressive disease; 2 from an infection; 1 from a second neoplasia; 7 from causes unrelated to therapy). Follow-up data regarding PFS and overall survival are not sufficiently mature to be analyzed.
Common grade 3 or 4 adverse events were fatigue (18.9%), anemia (17%), neutropenia (16.5%), lymphocytopenia (15.9%), and pneumonia (15.9%). Infusion reactions occurred in 13 patients (7.2%) and were always of grade 1 or 2. Infusion reactions resolved in all patients and no case discontinued treatment.
Conclusions
Our real world preliminary data confirm that EloRd is an effective and safe regimen for RRMM patients, particularly if used as first salvage regimen, basically resembling results obtained in controlled clinical trials.
Galli:Celgene: Honoraria; Janssen: Honoraria; Sigma-Tau: Honoraria; Bristol-Myers Squibb: Honoraria. Giuliani:Janssen Pharmaceutica: Other: Avisory Board, Research Funding; Celgene Italy: Other: Avisory Board, Research Funding; Takeda Pharmaceutical Co: Research Funding. Mangiacavalli:Janssen: Consultancy; Celgene: Consultancy; Cilag: Consultancy. Ballanti:Celgene: Honoraria; Janssen: Honoraria; Amgen: Honoraria; BMS: Honoraria. Montefusco:Celgene: Other: Advisory Board; Amgen: Other: Advisory Board; Janssen: Other: Advisory Board. Bringhen:Bristol-Myers Squibb: Honoraria; Celgene: Honoraria; Amgen: Honoraria, Other: Advisory Board; Janssen: Honoraria, Other: Advisory Board; Takeda: Consultancy. Zamagni:Amgen: Honoraria, Membership on an entity's Board of Directors or advisory committees; Celgene: Honoraria, Membership on an entity's Board of Directors or advisory committees; Janssen: Honoraria, Membership on an entity's Board of Directors or advisory committees; Takeda: Honoraria, Membership on an entity's Board of Directors or advisory committees; BMS: Honoraria, Membership on an entity's Board of Directors or advisory committees. Patriarca:Celgene: Other: Advisory Role; Travel, accommodations, expenses; Janssen: Other: Advisory role; Jazz: Other: Travel, accommodations, expenses; Medac: Other: Travel, accommodations, expenses; MSD Italy: Other: Advisory Role. Di Raimondo:Takeda: Honoraria, Research Funding; Celgene: Honoraria. Petrucci:Takeda Oncology; Amgen; Celgene; BMS; Janssen Cilag: Honoraria, Other: Advisory Board. Offidani:Bristol-Myers Squibb: Honoraria, Other: Advisory Board; Amgen: Honoraria, Other: Advisory Board; Takeda: Honoraria, Other: Advisory Board; Janssen: Honoraria, Other: Advisory Board; Celgene: Honoraria, Other: Advisory Board.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal